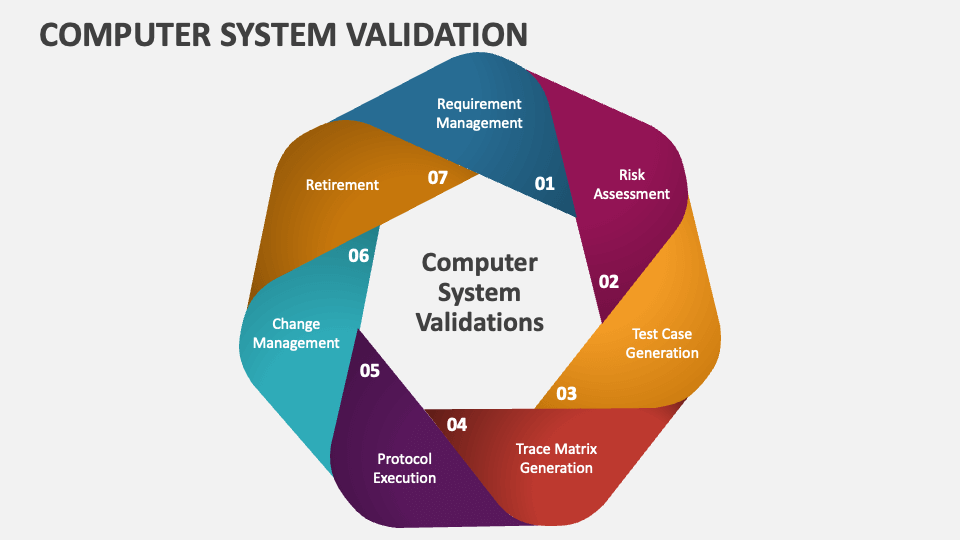
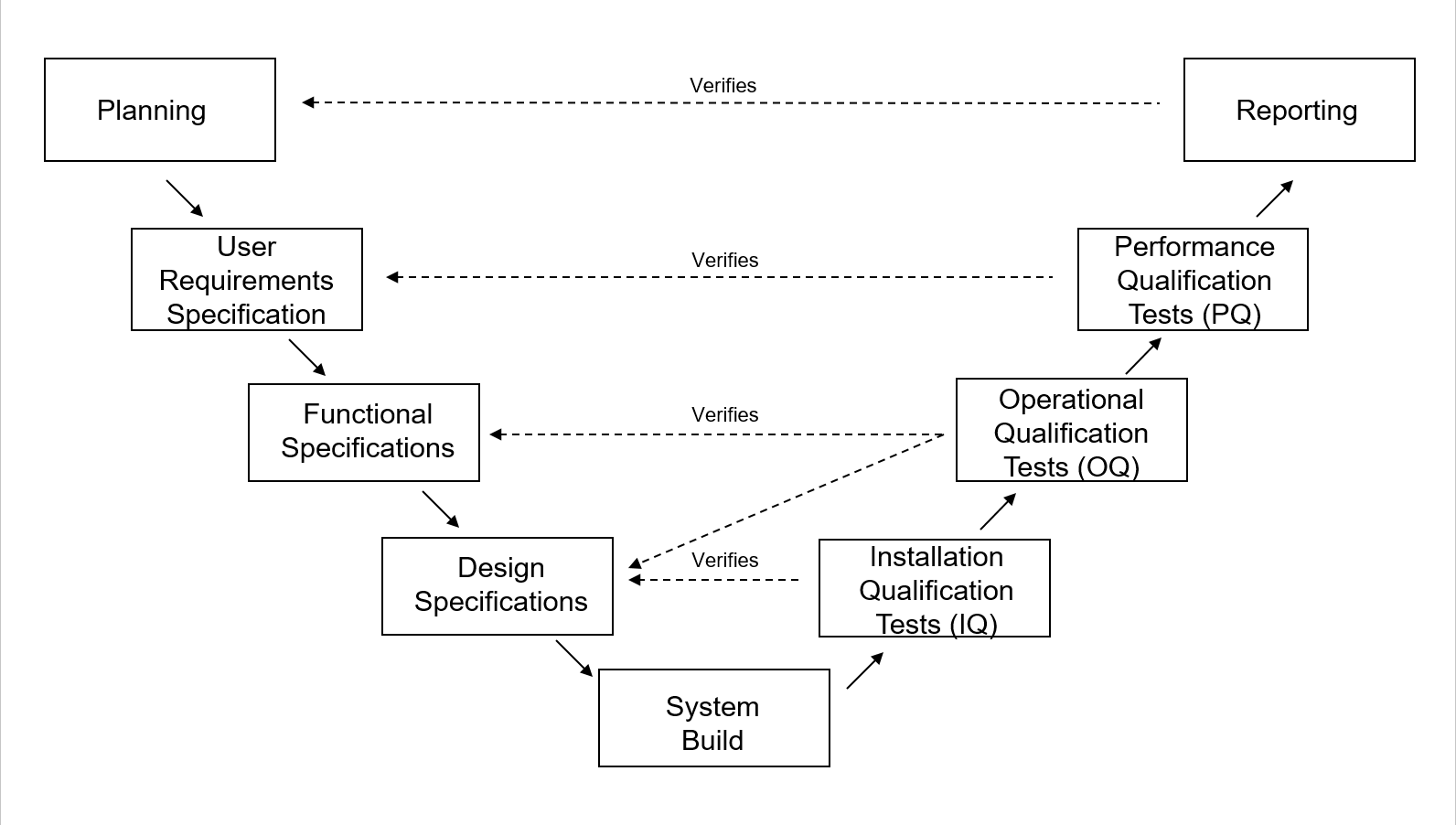
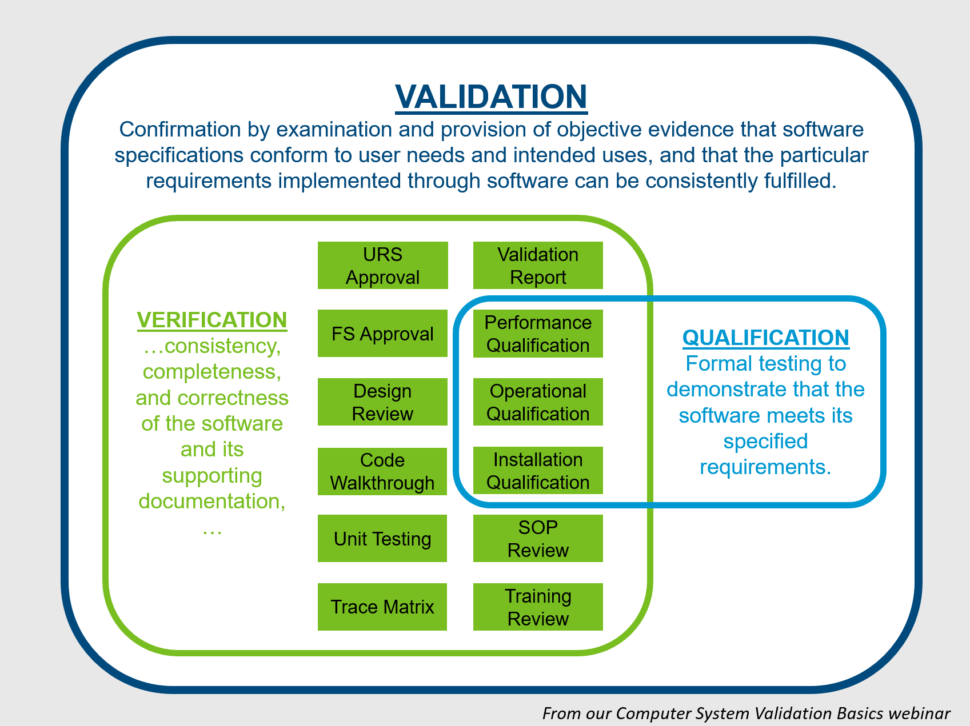
Computer System Validation Courses
Computer System Validation Courses - Earn a gxp computer system validation certificate. Understand how to ensure that software meets requirements and performs as expected. The instructor reviews recent fda. Where does validation fit into your sdlc or salc? Help your people gain a fuller understanding of the requirements for computer system validation and explore the methods to comply with them with our fda computer system validation. Understand concepts such as system categorization,. Validation master plan and system. Helping you become a certified csv. Start by gaining fundamental knowledge about computer system validation and maintenance based on the gamp 5 second edition. Document your dedication to computer system validation (csv) by earning a professional certification from biopharma institute. Which systems do you need to validate? Document your dedication to computer system validation (csv) by earning a professional certification from biopharma institute. Upon completion of this course you will be able to, understand the importance of computer system validation, the regulations and standards behind csv, recommendations and best practices. Understand how to ensure that software meets requirements and performs as expected. The instructor reviews recent fda. Pharmout has been training on computer systems validation since 2008, much has changed recently, surprisingly data integrity remains a hot topic with regulators. Understand concepts such as system categorization,. This course is designed for validation, quality, it, and business personnel responsible for implementing and using regulated computer systems in the pharmaceutical, biotech and. Learn verification and validation techniques for software quality assurance. Helping you become a certified csv. Start by gaining fundamental knowledge about computer system validation and maintenance based on the gamp 5 second edition. Pharmout has been training on computer systems validation since 2008, much has changed recently, surprisingly data integrity remains a hot topic with regulators. Computer system validation for professionals program includes an introduction to the international principles and regulations behind effective validation and. Understand concepts such as system categorization,. Document your dedication to computer system validation (csv) by earning a professional certification from biopharma institute. Understand how to ensure that software meets requirements and performs as expected. In this computer system validation training you’ll learn how to integrate risk based supplier evaluation into the computer system validation process. Which systems do you need. Validation master plan and system. Which systems do you need to validate? Pharmout has been training on computer systems validation since 2008, much has changed recently, surprisingly data integrity remains a hot topic with regulators. Upon completion of this course you will be able to, understand the importance of computer system validation, the regulations and standards behind csv, recommendations and. The instructor reviews recent fda. Document your dedication to computer system validation (csv) by earning a professional certification from biopharma institute. Where does validation fit into your sdlc or salc? These are some of the questions answered in this live. Helping you become a certified csv. What is computer system validation? In this computer system validation training you’ll learn how to integrate risk based supplier evaluation into the computer system validation process. This course is designed for validation, quality, it, and business personnel responsible for implementing and using regulated computer systems in the pharmaceutical, biotech and. Csv training, a subsidiary of validation associates llc, specializes in. Pharmout has been training on computer systems validation since 2008, much has changed recently, surprisingly data integrity remains a hot topic with regulators. Help your people gain a fuller understanding of the requirements for computer system validation and explore the methods to comply with them with our fda computer system validation. Helping you become a certified csv. Upon completion of. Learn verification and validation techniques for software quality assurance. Start by gaining fundamental knowledge about computer system validation and maintenance based on the gamp 5 second edition. Earn a gxp computer system validation certificate. Validation master plan and system. Upon completion of this course you will be able to, understand the importance of computer system validation, the regulations and standards. The instructor reviews recent fda. Understand how to ensure that software meets requirements and performs as expected. Pharmout has been training on computer systems validation since 2008, much has changed recently, surprisingly data integrity remains a hot topic with regulators. In this computer system validation training you’ll learn how to integrate risk based supplier evaluation into the computer system validation. This course is designed for validation, quality, it, and business personnel responsible for implementing and using regulated computer systems in the pharmaceutical, biotech and. Earn a gxp computer system validation certificate. Csv training, a subsidiary of validation associates llc, specializes in software validation, cleaning validation, instrument/equipment validation, process validation, documentum,. These are some of the questions answered in this live.. What is computer system validation? Understand concepts such as system categorization,. Which systems do you need to validate? This course is designed for validation, quality, it, and business personnel responsible for implementing and using regulated computer systems in the pharmaceutical, biotech and. Validation master plan and system. The instructor reviews recent fda. Learn verification and validation techniques for software quality assurance. Understand concepts such as system categorization,. These are some of the questions answered in this live. Validation master plan and system. This course is designed for validation, quality, it, and business personnel responsible for implementing and using regulated computer systems in the pharmaceutical, biotech and. Earn a gxp computer system validation certificate. Understand how to ensure that software meets requirements and performs as expected. Computer system validation for professionals program includes an introduction to the international principles and regulations behind effective validation and qualification. Helping you become a certified csv. Csv training, a subsidiary of validation associates llc, specializes in software validation, cleaning validation, instrument/equipment validation, process validation, documentum,. What is computer system validation? Start by gaining fundamental knowledge about computer system validation and maintenance based on the gamp 5 second edition. The course will equip you with recommendations and best practices for successful csv implementation, as well as an understanding of the gamp®5 framework and the essential. Which systems do you need to validate? Pharmout has been training on computer systems validation since 2008, much has changed recently, surprisingly data integrity remains a hot topic with regulators.Computer System Validation (CSV & CSA) LearnGxP Accredited Online
Computer System Validation (CSV) Online Course And Certification
Computer System Validation Course AnumTechSystems, Inc.
Computer System Validation (CSV) Validation Testing Part 1 LearnGxP
Computer System Validation PowerPoint and Google Slides Template PPT
What is Computer System Validation and How Do You Do It?
What is Computer System Validation and How Do You Do It?
Computer System Validation (CSV) Basic Concepts, Regulations and GAMP
Computer System Validation Training Course (Recorded)
Computer System Validation (CSV) Validation Testing Part 1 LearnGxP
In This Computer System Validation Training You’ll Learn How To Integrate Risk Based Supplier Evaluation Into The Computer System Validation Process.
Where Does Validation Fit Into Your Sdlc Or Salc?
Document Your Dedication To Computer System Validation (Csv) By Earning A Professional Certification From Biopharma Institute.
Upon Completion Of This Course You Will Be Able To, Understand The Importance Of Computer System Validation, The Regulations And Standards Behind Csv, Recommendations And Best Practices.
Related Post: