Good Manufacturing Practice Online Course
Good Manufacturing Practice Online Course - This course is an introduction to the principles and practices of good manufacturing practices (gmp). The digital transformation of manufacturing needs a workforce highly skilled in digital technologies to meet the growing demands of modern manufacturing. Fill in the form below 👇🏼. Pharmaceutical manufacturers must comply with the regulatory requirements of the countries where they produce and sell medical products. Elevate your leadership, amplify your communication skills 1 may,. Guide to current good manufacturing practices (cgmp) 1 may, 2025; A comprehensive guide in today’s rapidly evolving pharmaceutical landscape, maintaining compliance with good manufacturing. Affordable, online training to equip frontline workers. Good manufacturing practice course online: Accredited good manufacturing practices trainings designed for life sciences. Pharmaceutical manufacturers must comply with the regulatory requirements of the countries where they produce and sell medical products. This free online course provides an overview of essential gmp principles and requirements. Global leadership, lean principles for. Learn the core principles of good manufacturing practices and their importance in ensuring the safety and quality of pharmaceutical products. Good manufacturing practice course online: Our course is accredited by the international haccp alliance and recognized by fda, usda and gfsi. Affordable, online training to equip frontline workers. Let's explore how to design and deliver online employee safety training in manufacturing. The digital transformation of manufacturing needs a workforce highly skilled in digital technologies to meet the growing demands of modern manufacturing. Equip workers with expertly curated manufacturing training from industry leaders. It covers key principles of the who and pic/s standards and will equip participants with a. Let's explore how to design and deliver online employee safety training in manufacturing. Welcome to the certified pharmaceutical gmp professional course, meticulously crafted by cdg to equip professionals with the essential knowledge and skills required to excel in the. Accredited good manufacturing practices trainings. A comprehensive guide in today’s rapidly evolving pharmaceutical landscape, maintaining compliance with good manufacturing. Affordable, online training to equip frontline workers. The european medicines agency's (ema) provides answers to frequently asked questions on good manufacturing practice (gmp) and good distribution practice (gdp), as. Let's explore how to design and deliver online employee safety training in manufacturing. Reduce your risk of. This course is an introduction to the principles and practices of good manufacturing practices (gmp). The digital transformation of manufacturing needs a workforce highly skilled in digital technologies to meet the growing demands of modern manufacturing. Welcome to the certified pharmaceutical gmp professional course, meticulously crafted by cdg to equip professionals with the essential knowledge and skills required to excel. Equip workers with expertly curated manufacturing training from industry leaders. Elevate your leadership, amplify your communication skills 1 may,. Reduce your risk of recalls from foreign material and protect your brand. Good manufacturing practice course online: Get an individual certification for your gmp skills. Stay ahead of regulatory requirements. Welcome to the certified pharmaceutical gmp professional course, meticulously crafted by cdg to equip professionals with the essential knowledge and skills required to excel in the. Guide to current good manufacturing practices (cgmp) 1 may, 2025; The european medicines agency's (ema) provides answers to frequently asked questions on good manufacturing practice (gmp) and good distribution. A comprehensive guide in today’s rapidly evolving pharmaceutical landscape, maintaining compliance with good manufacturing. Good manufacturing practice course online: The digital transformation of manufacturing needs a workforce highly skilled in digital technologies to meet the growing demands of modern manufacturing. Elevate your leadership, amplify your communication skills 1 may,. Learn the core principles of good manufacturing practices and their importance. The digital transformation of manufacturing needs a workforce highly skilled in digital technologies to meet the growing demands of modern manufacturing. Fill in the form below 👇🏼. Let's explore how to design and deliver online employee safety training in manufacturing. Welcome to the certified pharmaceutical gmp professional course, meticulously crafted by cdg to equip professionals with the essential knowledge and. Welcome to the certified pharmaceutical gmp professional course, meticulously crafted by cdg to equip professionals with the essential knowledge and skills required to excel in the. Pharmaceutical manufacturers must comply with the regulatory requirements of the countries where they produce and sell medical products. Stay ahead of regulatory requirements. A comprehensive guide in today’s rapidly evolving pharmaceutical landscape, maintaining compliance. Elevate your leadership, amplify your communication skills 1 may,. Let's explore how to design and deliver online employee safety training in manufacturing. Contact us · managers · manufacturing Reduce your risk of recalls from foreign material and protect your brand. Accredited good manufacturing practices trainings designed for life sciences. It requires strategic planning, engaging content, and powerful training software. Guide to current good manufacturing practices (cgmp) 1 may, 2025; Stay ahead of regulatory requirements. This course is an introduction to the principles and practices of good manufacturing practices (gmp). Reduce your risk of recalls from foreign material and protect your brand. A comprehensive guide in today’s rapidly evolving pharmaceutical landscape, maintaining compliance with good manufacturing. Pharmaceutical manufacturers must comply with the regulatory requirements of the countries where they produce and sell medical products. Contact us · managers · manufacturing Learn the core principles of good manufacturing practices and their importance in ensuring the safety and quality of pharmaceutical products. It covers key principles of the who and pic/s standards and will equip participants with a. The european medicines agency's (ema) provides answers to frequently asked questions on good manufacturing practice (gmp) and good distribution practice (gdp), as. Welcome to the certified pharmaceutical gmp professional course, meticulously crafted by cdg to equip professionals with the essential knowledge and skills required to excel in the. Get an individual certification for your gmp skills. Good manufacturing practice course online: Our course is accredited by the international haccp alliance and recognized by fda, usda and gfsi. Accredited good manufacturing practices trainings designed for life sciences. Elevate your leadership, amplify your communication skills 1 may,. Affordable, online training to equip frontline workers. Whether it's safety essentials or advanced skills. This is 2 day (16 hours) course offered in our training center in chicagoland. Guide to current good manufacturing practices (cgmp) 1 may, 2025;Introducing Our New Good Manufacturing Practice (GMP) Online Course
GMP Training Online Course
5 Key Elements of Good Manufacturing Practices LearnGxP Accredited
Good Manufacturing Practice (GMP) in EU Online Certified Course
Good Manufacturing Practices (GMP) Online Course (Recorded)
Good Manufacturing Practices Course Institute for SmallScale Industries
Basic Good Manufacturing Practices Guidelines GMP Certification Online
Good Manufacturing Practice Training John Academy
Good Manufacturing Practice (GMP) Alpha Academy
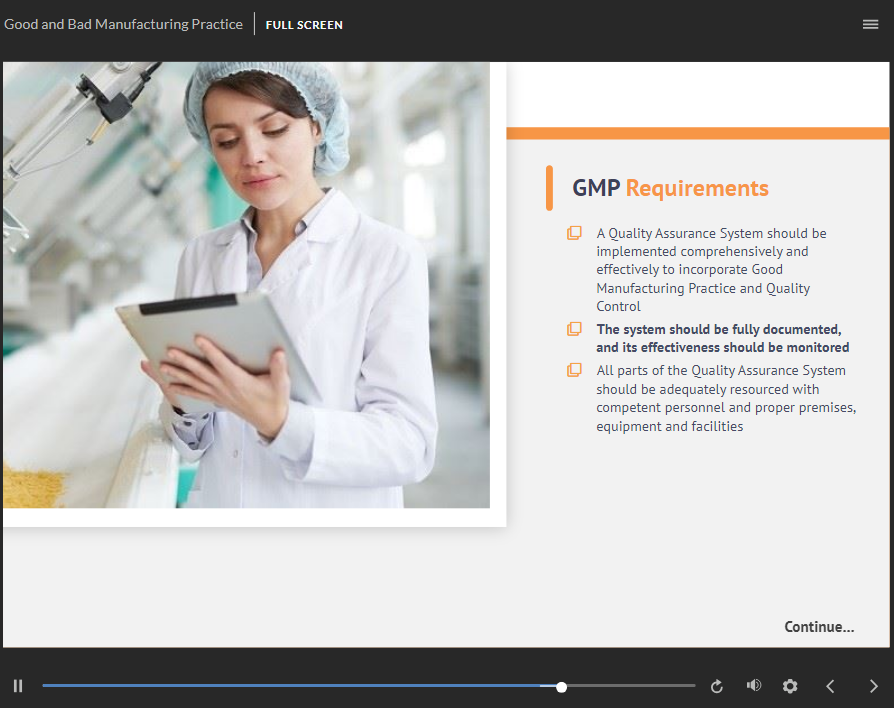
Good Manufacturing Practice 01 GMP Online Training Part 1
Reduce Your Risk Of Recalls From Foreign Material And Protect Your Brand.
Stay Ahead Of Regulatory Requirements.
The Digital Transformation Of Manufacturing Needs A Workforce Highly Skilled In Digital Technologies To Meet The Growing Demands Of Modern Manufacturing.
Equip Workers With Expertly Curated Manufacturing Training From Industry Leaders.
Related Post: