Software Validation Course
Software Validation Course - Software testing and validation are vital because they ensure that the end product meets the design requirements. Understand how to ensure that software meets requirements and performs as expected. In just two weeks, you'll gain a comprehensive understanding of industry. Courses include voiceovers, easy navigation, reading materials, case. Gain insights into fda regulations with fda software validation guidelines. Training is current with respect to regulations, procedures, and 3rd party validated and/or accredited. This course covers the principles of software validation, computer systems validation and requirements for electronic records and electronic signatures. Software verification and validation (v&v) course by software excellence academy is designed to provide a knowledge base and practical skills for anyone interested in implementing or. Document your dedication to regulations, gxp compliance, quality, consumer safety, and job performance by earning a professional certification from biopharma institute. Helping you become a certified csv. Training is current with respect to regulations, procedures, and 3rd party validated and/or accredited. This course will help you understand software verification and validation and the impact of these new revisions on software used by medical device manufacturers. In just two weeks, you'll gain a comprehensive understanding of industry. Fda software validation guidelines training. Document your dedication to regulations, gxp compliance, quality, consumer safety, and job performance by earning a professional certification from biopharma institute. This course covers the principles of software validation, computer systems validation and requirements for electronic records and electronic signatures. Helping you become a certified csv. Gain insights into fda regulations with fda software validation guidelines. The “fundamentals of software testing & validation training” by tonex provides a comprehensive overview of essential software testing and validation techniques. Software verification and validation (v&v) course by software excellence academy is designed to provide a knowledge base and practical skills for anyone interested in implementing or. Software verification and validation (v&v) course by software excellence academy is designed to provide a knowledge base and practical skills for anyone interested in implementing or. Software testing and validation are vital because they ensure that the end product meets the design requirements. Gain insights into fda regulations with fda software validation guidelines. Helping you become a certified csv. The. Fda software validation guidelines training by tonex. Software testing and validation are vital because they ensure that the end product meets the design requirements. Fda software validation guidelines training. This course covers the principles of software validation, computer systems validation and requirements for electronic records and electronic signatures. Software verification and validation (v&v) course by software excellence academy is designed. Understand how to ensure that software meets requirements and performs as expected. Helping you become a certified csv. Gain insights into fda regulations with fda software validation guidelines. Software testing and validation are vital because they ensure that the end product meets the design requirements. Learn verification and validation techniques for software quality assurance. Trusted certificationaccredited online coursescpd / ceu accreditation Course objectives include the following: This course covers the principles of software validation, computer systems validation and requirements for electronic records and electronic signatures. Understand how to ensure that software meets requirements and performs as expected. Courses include voiceovers, easy navigation, reading materials, case. Fda software validation guidelines training. Course objectives include the following: Courses include voiceovers, easy navigation, reading materials, case. Understand how to ensure that software meets requirements and performs as expected. We know there is often a lack of training funds or time in your schedule to attend full days of training. Training is current with respect to regulations, procedures, and 3rd party validated and/or accredited. This course covers the principles of software validation, computer systems validation and requirements for electronic records and electronic signatures. Document your dedication to regulations, gxp compliance, quality, consumer safety, and job performance by earning a professional certification from biopharma institute. The “fundamentals of software testing &. Fda software validation guidelines training by tonex. Helping you become a certified csv. Gain insights into fda regulations with fda software validation guidelines. Trusted certificationaccredited online coursescpd / ceu accreditation Software testing and validation are vital because they ensure that the end product meets the design requirements. Software testing and validation are vital because they ensure that the end product meets the design requirements. Helping you become a certified csv. Courses include voiceovers, easy navigation, reading materials, case. The “fundamentals of software testing & validation training” by tonex provides a comprehensive overview of essential software testing and validation techniques. Course objectives include the following: Training is current with respect to regulations, procedures, and 3rd party validated and/or accredited. In just two weeks, you'll gain a comprehensive understanding of industry. Helping you become a certified csv. The “fundamentals of software testing & validation training” by tonex provides a comprehensive overview of essential software testing and validation techniques. Software verification and validation (v&v) course by software. Understand how to ensure that software meets requirements and performs as expected. Software verification and validation (v&v) course by software excellence academy is designed to provide a knowledge base and practical skills for anyone interested in implementing or. Course objectives include the following: Training is current with respect to regulations, procedures, and 3rd party validated and/or accredited. Gain insights into. Software verification and validation (v&v) course by software excellence academy is designed to provide a knowledge base and practical skills for anyone interested in implementing or. This course covers the principles of software validation, computer systems validation and requirements for electronic records and electronic signatures. Helping you become a certified csv. We know there is often a lack of training funds or time in your schedule to attend full days of training. Training is current with respect to regulations, procedures, and 3rd party validated and/or accredited. Learn verification and validation techniques for software quality assurance. Understand how to ensure that software meets requirements and performs as expected. Trusted certificationaccredited online coursescpd / ceu accreditation Document your dedication to regulations, gxp compliance, quality, consumer safety, and job performance by earning a professional certification from biopharma institute. In just two weeks, you'll gain a comprehensive understanding of industry. Gain insights into fda regulations with fda software validation guidelines. This course will help you understand software verification and validation and the impact of these new revisions on software used by medical device manufacturers. The “fundamentals of software testing & validation training” by tonex provides a comprehensive overview of essential software testing and validation techniques. Enhance your skills without leaving your office. Fda software validation guidelines training by tonex.Software Verification, Validation and Compliance. V Shaped Model
Software Validation (CSV) Online Course YouTube
Software Validation
Software Validation Online Course YouTube
Software Validation Online Course And Certification
PPT SWE 205 Introduction to Software Engineering PowerPoint
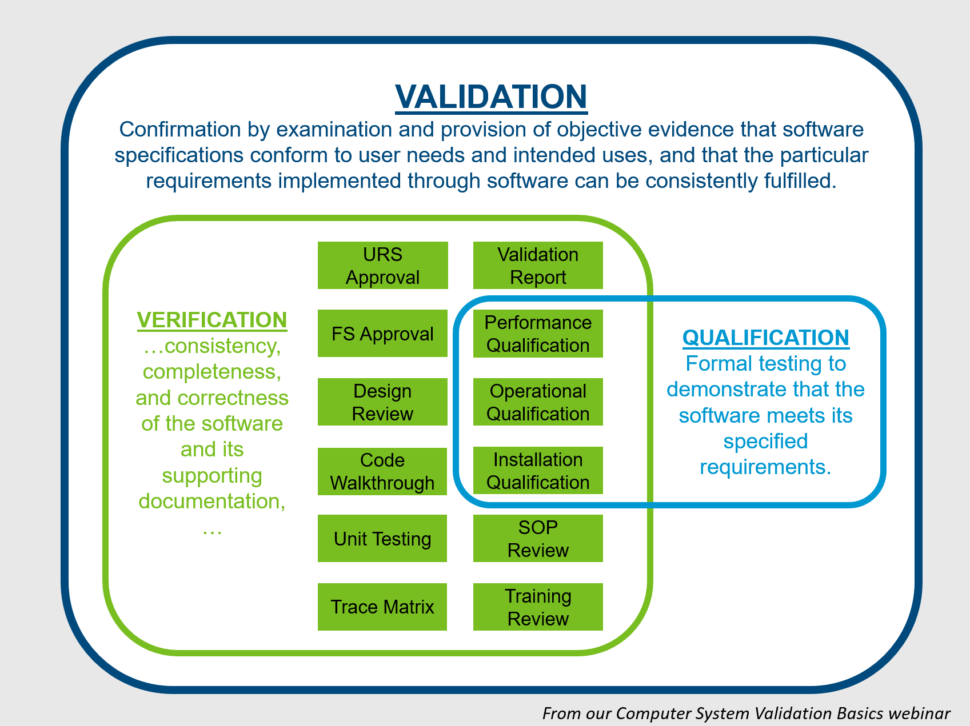
What is Computer System Validation and How Do You Do It?
Software Validation Online Training YouTube
Software Validation Full Details PresentationEZE
What are the Software Validation Requirements of ISO 134852016
Software Testing And Validation Are Vital Because They Ensure That The End Product Meets The Design Requirements.
Course Objectives Include The Following:
Fda Software Validation Guidelines Training.
Courses Include Voiceovers, Easy Navigation, Reading Materials, Case.
Related Post: